1. Introduction
Recently, a new consensus statement has been published by a group of UK scientific societies regarding the perioperative management of adult patients taking GLP‑1 receptor agonists (GLP‑1 RAs), GIP agonists, and SGLT2 inhibitors. I thought it would be timely to write an article about this topic—especially since, as anesthesiologists, we are increasingly encountering patients on these medications who are undergoing surgery.
2. GLP‑1 RAs and GIP Agonists
2.1 Mechanism of Action
GLP‑1 and GIP are incretins secreted by specialized cells in the intestinal tract. Specifically, GLP‑1 is secreted by L cells and GIP by K cells in response to the presence of glucose in the gut lumen. Their key actions include:
- Insulin Secretion: Both GLP‑1 and GIP enhance insulin secretion from pancreatic beta cells.
- Glucagon Modulation: GLP‑1 reduces glucagon secretion, whereas GIP tends to increase it.
- Gastric Emptying: Both delay gastric emptying, which results in a slower rise in postprandial blood glucose.
- Satiety and Nausea: Both activate satiety centers in the hypothalamus, which may contribute to the nausea commonly seen during drug initiation or dose escalation.
- Insulin Sensitivity: Both agents improve insulin sensitivity.

2.2 Indications
The currently approved indications for these agents include:
- Type 2 diabetes mellitus
- Overweight and obesity
Beyond diabetes and obesity, further indications under investigation for GLP‑1 RAs include:
- Osteoarthritis
- Obstructive sleep apnea
- Metabolic dysfunction-associated steatotic liver disease
- Metabolic dysfunction-associated steatohepatitis
2.3 Benefits and Harms of Continuation/Discontinuation in the Perioperative Period:
2.3.1 Benefits:
According to the guidelines, the acute use of these drugs may offer benefits based on published data:
- Improved perioperative glycemic control
- Lower postoperative insulin requirements
- Reduced risk of hypoglycemia
- These benefits may lead to reduced complications associated with hyperglycemia (such as a shorter hospital stay, fewer surgical site infections, less acute kidney injury, fewer acute coronary syndromes, reduced critical care admissions, and shorter durations on a ventilator)
- Improved cardiovascular outcomes
2.3.2 Harms
- There is an increased risk of pulmonary aspiration. In one study, the odds ratio (95% CI) was 10.23 (2.94–35.82) in the elective surgical setting, and the hazard ratio (95% CI) was 1.33 (1.02–1.74) in the endoscopy setting. This risk may be explained by the delay in gastric emptying caused by incretins and the associated increase in nausea; however, the effect of tachyphylaxis on gastric emptying (especially with chronic use) remains unclear.
- GLP‑1 RAs commonly induce side effects such as nausea, vomiting, abdominal pain, and constipation. These symptoms may complicate the diagnosis and management of perioperative conditions that share similar clinical presentations. The incidence of these side effects during chronic use is not yet clearly defined.
2.4 What Do the Guidelines Say?
"Box 1. Recommendations for the peri-operative management of patients taking glucagon-like peptide-1 receptor agonists and glucose-dependent insulinotropic peptide [1] :
• The risk of pulmonary aspiration and mitigation strategies should be discussed with the patient using a shared decision-making approach."
• "Patients should continue to take glucagon-like peptide-1 receptor agonists throughout the perioperative period."
• "Patients and clinicians should adhere to recommended fasting guidelines."
• "Upper gastrointestinal symptoms alone should not be used to determine gastric content."
• "Regional anaesthesia should be considered as the primary anaesthetic technique, if appropriate."
• "Point-of-care gastric ultrasound should be considered before induction of anaesthesia to facilitate risk stratification, if appropriate."
• "Individualised pulmonary aspiration risk assessment should be completed, accounting for drug, patient and procedural factors."
• "Anaesthesia and airway management should aim to reduce the risk of pulmonary aspiration on induction of anaesthesia, during maintenance and after emergence of anaesthesia. This could include: administering prokinetics; using a tracheal tube; modified rapid sequence intubation (with or without cricoid force, depending on local practice); head-up position for induction of anaesthesia; potential use of gastric tubes to empty the stomach before induction of anaesthesia and tracheal extubation; and awake tracheal extubation."
2.5 Comparison with Other Guidelines:
2.5.1 ASA Guidelines:
A multisociety consensus for the perioperative management of patients taking GLP‑1 RAs was developed by representatives from:
- The American Society of Anesthesiologists
- The American Gastroenterological Association
- The American Society for Metabolic and Bariatric Surgery
- The International Society of Perioperative Care of Patients with Obesity
- The Society of American Gastrointestinal and Endoscopic Surgeons
Recommendation 1[2] :
Consider variables that increase the risk of delayed gastric emptying and aspiration with the periprocedural use of GLP-1RA:- The escalation phase of the medication- Higher doses- Weekly dosing- The presence of gastrointestinal symptoms (nausea, vomiting, abdominal pain, dyspepsia, and constipation) suggestive of delayed gastric emptying- Other medical conditions that may delay gastric emptying.
GLP‑1 RA therapy may be continued preoperatively in patients without an elevated risk of delayed gastric emptying and aspiration. If the decision is made to hold GLP‑1 RAs, follow ASA guidance: hold daily formulations on the day of surgery and weekly formulations one week prior. Assess all patients for symptoms of delayed gastric emptying on the day of the procedure[2] [8].
Recommendation 2[2]:
- Minimize the aspiration risk by modifying the preoperative diet and/or altering the anesthesia plan.- Consider a preoperative diet modification (e.g., a liquid diet for at least 24 hours) for patients with concerns about delayed gastric emptying.- Use point-of-care gastric ultrasound to assess aspiration risk if there is clinical concern for retained gastric contents.- If there is a clinical concern for retained gastric contents, weigh the benefits and risks of rapid sequence induction of general anesthesia versus procedure cancellation.
N.B: the formulation of these two recommendations has been changed!
2.5.2 ESAIC Guidelines:
The ESAIC has published new guidelines on the preoperative assessment of adults undergoing elective noncardiac surgery, dedicating a full section to the perioperative management of GLP‑1 RAs and SGLT2 inhibitors[3]:
"R12.1: When a GLP-1 agonist is prescribed as a weekly injection and considering the long half-life of GLP-1 agonists, we recommend pausing GLP-1 agonists at least 1 week before a scheduled procedure requiring sedation/anaesthesia. If these drugs are given for obesity, then 2 weeks (three half-lives) are recommended. (CPS)"
"R12.2: If the medication is prescribed as daily oral or subcutaneous administration, we recommend pausing GLP-1 agonists on the day of the procedure. (CPS)"
"R12.3: There is no evidence to show that stopping these medications even 1 week before the procedure will eliminate the risk of delayed gastric emptying, despite following the usual fasting timing for surgery. (CPS)"
"R12.4: A clear fluid diet should be encouraged 24 h before any procedure in patients taking GLP-1 agonists. (CPS)"
"R12.5: All patients taking GLP-1 agonists should be considered as at risk of having a full stomach despite a lack of gastrointestinal symptoms. (CPS)"
"R12.6: Whenever possible, a gastric ultrasound should be performed. If gastric contents are found by ultrasound and these are considered as a high risk for aspiration, patients should be counselled about this risk before deciding to proceed with sedation/general anaesthesia. (CPS)"
"R12.7: If the procedure is of such urgency that postponement is not desirable, endotracheal intubation by rapid sequence induction/intubation is advised.(CPS)"
3. SGLT2 Inhibitors (SGLT2i):
3.1 Mechanism of Action:
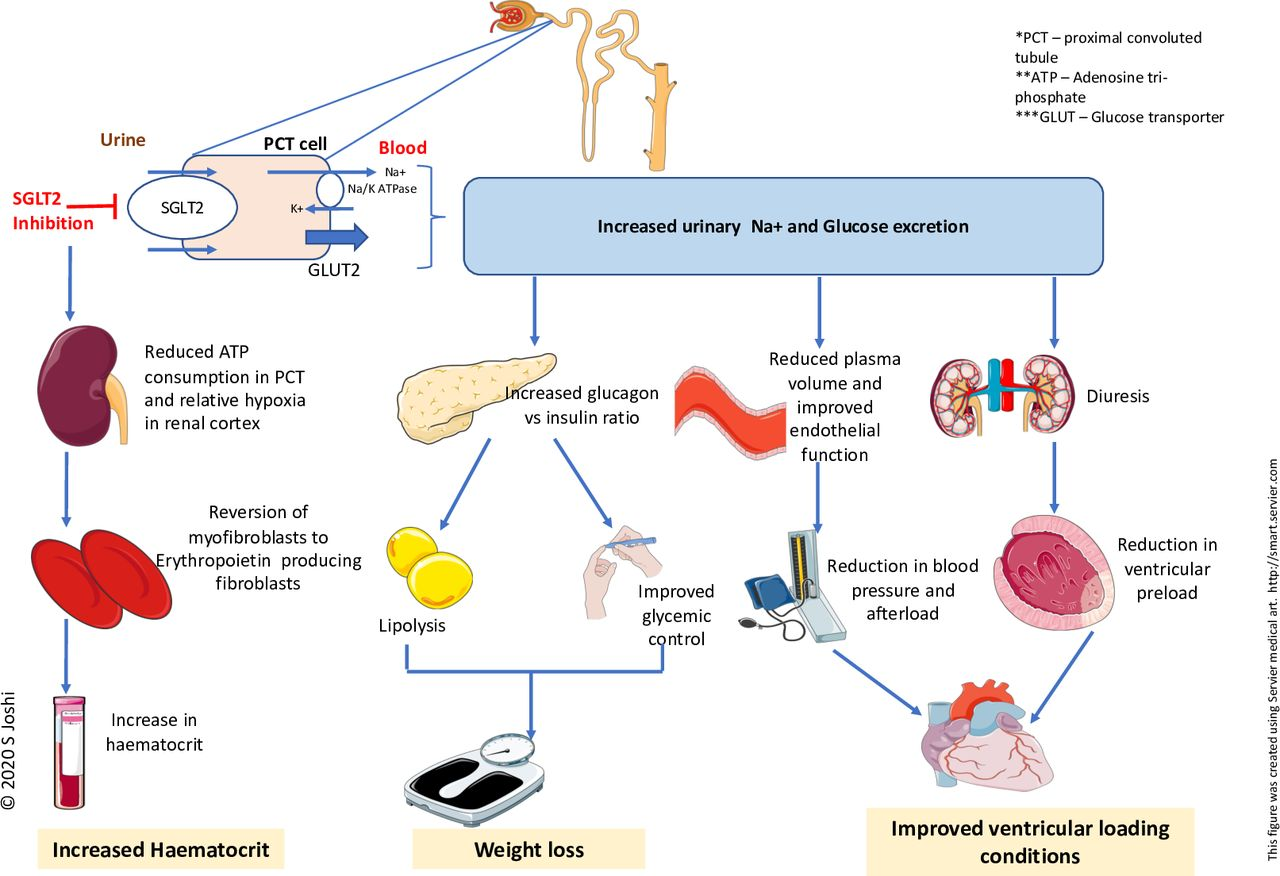
SGLT2 inhibitors (commonly known as “-flozin” drugs) work by blocking the SGLT2 protein in the proximal tubule of the kidneys. This protein is responsible for reabsorbing filtered sodium and glucose; blocking it leads to reduced blood glucose levels and increased sodium excretion.
3.2 Indications:
The approved indications for SGLT2 inhibitors include:
- Diabetes mellitus
- Heart failure
- Chronic kidney disease
3.3 Benefits and Harms of Continuation/Discontinuation in the Perioperative Period:
3.3.1 Benefits:
- Improved Glycemic Control: Continued use may help maintain stable blood glucose levels.
- Heart Failure Patients: Data suggest that discontinuing SGLT2 inhibitors in patients treated for heart failure may be associated with worsening heart failure, so maintaining treatment may be beneficial.
3.3.2 Harms:
- Risk of Ketoacidosis: The major concern is the development of diabetic ketoacidosis (DKA), including euglycemic DKA. The inhibition of glucose reabsorption increases ketone production, and the stress of the perioperative period may further predispose patients to hyperketonemia. One study reported a higher risk of perioperative DKA in patients taking SGLT2 inhibitors (1.02 vs. 0.69 per 1000 patients, OR 1.48; 95% CI 1.02–2.15; p=0.037)[1].
- Hyperglycemia: Ceasing SGLT2 inhibitors may lead to hyperglycemia and its associated complications.
- Heart Failure: In patients with heart failure, stopping SGLT2 inhibitors may be associated with worsening of the condition.
3.4 What Do the Guidelines Say?
"Box 2. Recommendations for the peri-operative management of patients taking sodium-glucose cotransporter-2 inhibitors [1]:
• The risk of peri-operative ketoacidosis and mitigation strategies should be discussed with the patient using a shared decision-making approach."
•"Sodium-glucose cotransporter-2 inhibitors should be omitted the day before and the day of a procedure. "
•"Patients and clinicians should adhere to recommended fasting guidelines and avoid prolonged starvation times."
•"For patients discharged from hospital on the day of surgery, sodium-glucose cotransporter-2 inhibitors should be restarted once eating and drinking normally (usually 24–48 h after surgery)."
•"For patients staying in hospital after surgery, consider restarting sodium-glucose cotransporter-2 inhibitors once eating and drinking normally and capillary ketones are < 0.6 mmol.l-1. "
•"For patients diagnosed with diabetes mellitus on a very low energy/liver reduction diet for the purposes of surgery, sodium-glucose cotransporter-2 inhibitors should be stopped at commencement of the diet, and adjust diabetes mellitus treatment as necessary."
•"Written sick-day rules should be provided to patients at pre-operative assessment and at discharge."
3.5 Comparison with Other Guidelines:
3.5.1 American Heart Association (AHA) Guidelines[4] :
"In patients scheduled for NCS, SGLT2i should be discontinued 3 to 4 days* days before surgery to reduce the risk of perioperative metabolic acidosis (COR I/LOE C-LD) *Canagliflozin, dapagliflozin, and empagliflozin should be stopped ≥3 days and ertugliflozin ≥4 days before scheduled surgery."
3.5.2 European Society of Cardiology (ESC) Guidelines[5] :
"It should be considered to interrupt SGLT-2 inhibitor therapy for at least 3 days before intermediate- and high-risk NCS." (Class IIa/Level C)
3.5.3 ESAIC Guidelines[3] :
"R12.8: SGLT2 inhibitors (SGLT2i) drugs should be withheld for 3 to 4 days before elective procedures to reduce the risk of euglycemic diabetic ketoacidosis. (CPS)"
"R12.9: Patients taking SGLT2i medications should consume clear fluids approximately 2 h before the procedure to keep regular hydration. (CPS)"
"R12.10: Euglycemic diabetic ketoacidosis should be suspected in this category of patients, and blood b-hydroxybutyrate (BOHB) is a functional confirmatory test. (CPS)"
"R12.11: If a patient taking SGLT2i drugs did not discontinue the medication in time, dehydration caused by bowel preparation for endoscopy can increase the ketone levels, and the patient should be adequately hydrated before leaving the hospital. (CPS)"
In summary, all three guidelines agree that SGLT2 inhibitors should be discontinued for at least 3 days preoperatively, with the main concern being the prevention of DKA/euglycemic DKA and metabolic acidosis due to increased ketone production. Additionally, maintaining adequate hydration, avoiding prolonged starvation, and ensuring appropriate glucose and ketone monitoring are crucial.
4. Conclusion:
The perioperative management of patients taking incretin‐based therapies (GLP‑1 and GIP receptor agonists) and SGLT2 inhibitors is evolving as these drugs gain widespread use for diabetes, obesity, and cardio‐renal protection. Current consensus guidelines (in patients without elevated risk of delayed gastric emptying and aspiration) advocate for the continuation of GLP‑1 receptor agonists and GIP agonists in the perioperative period—coupled with careful risk assessment, adherence to fasting protocols, and possible dietary modifications—to mitigate the risks of delayed gastric emptying and pulmonary aspiration. In contrast, SGLT2 inhibitors should be withheld (typically at least one day preoperatively) to minimize the risk of euglycemic diabetic ketoacidosis. The current evidence base is limited by a predominance of retrospective studies and case reports. Therefore, further large-scale, prospective research is essential to refine these guidelines, ensure individualized patient safety, and improve overall perioperative outcomes. A multidisciplinary approach and shared decision-making remain key to balancing the benefits of these therapies with their potential risks during the perioperative period.
5. References:
- Elective peri‐operative management of adults taking glucagon‐like peptide‐1 receptor agonists, glucose‐dependent insulinotropic peptide agonists and sodium‐glucose cotransporter‐2 inhibitors: A multidisciplinary consensus statement: A consensus statement from the Association of Anaesthetists, Association of British Clinical Diabetologists, British Obesity and Metabolic Surgery Society, Centre for Perioperative Care, Joint British Diabetes Societies for Inpatient Care, Royal College of Anaesthetists, Society for Obesity and Bariatric Anaesthesia and UK Clinical Pharmacy Association. Anaesthesia, anae.16541.
- Multisociety clinical practice guidance for the safe use of glucagon-like peptide-1 receptor agonists in the perioperative period. Surgery for Obesity and Related Diseases, 20(12), 1183–1186.
- Preoperative assessment of adults undergoing elective noncardiac surgery: Updated guidelines from the European Society of Anaesthesiology and Intensive Care. European Journal of Anaesthesiology, 42(1), 1–35.
- 2024 AHA/ACC/ACS/ASNC/HRS/SCA/SCCT/SCMR/SVM Guideline for Perioperative Cardiovascular Management for Noncardiac Surgery: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation, 150(19).
- 2022 ESC Guidelines on cardiovascular assessment and management of patients undergoing non-cardiac surgery: Developed by the task force for cardiovascular assessment and management of patients undergoing non-cardiac surgery of the European Society of Cardiology (ESC) Endorsed by the European Society of Anaesthesiology and Intensive Care (ESAIC), European Heart Journal, Volume 43, Issue 39, 14 October 2022, Pages 3826–3924.
- Role of a Dual Glucose-Dependent Insulinotropic Peptide (GIP)/Glucagon-like Peptide-1 Receptor Agonist (Twincretin) in Glycemic Control: From Pathophysiology to Treatment. Life, 12(1), 29.
- Sodium-glucose co-transporter 2 inhibitor therapy: mechanisms of action in heart failureHeart 2021;107:1032-1038.
- American Society of Anesthesiologists Consensus-Based Guidance on Preoperative Management of Patients (Adults and Children) on Glucagon-Like Peptide-1 (GLP-1) Receptor Agonists.
Keywords:
Perioperative management, GLP‑1 receptor agonists, GIP agonists, SGLT2 inhibitors, Diabetic ketoacidosis, Aspiration risk, Glycemic control, Preoperative fasting, Obesity management, Anesthesia safety, Cardiorenal protection, Consensus guidelines, Shared decision-making, Perioperative complications.

Comments ()