1-Introduction:
Sepsis, a life-threatening condition arising from the body's dysregulated response to infection, requires prompt and effective treatment with antibiotics. However, determining the optimal duration of antibiotic therapy remains a challenge. Prolonged treatment can drive antibiotic resistance, increase adverse effects, and inflate healthcare costs, while prematurely discontinuing antibiotics risks treatment failure and worse outcomes.
A recent study, the Biomarker-Guided Duration of Antibiotic Treatment in Hospitalised Patients With Suspected Sepsis (ADAPT-Sepsis) trial, published in JAMA, sought to address this dilemma by evaluating the safety and effectiveness of using biomarkers to guide antibiotic discontinuation in critically ill patients with suspected sepsis.
2-Study Design: Rigorous and Patient-Centered
The ADAPT-Sepsis trial was a multicenter, randomized clinical trial conducted in 41 intensive care units across the UK, involving 2,760 adult patients. The study aimed to determine if using procalcitonin (PCT) or C-reactive protein (CRP) to guide the duration of antibiotic therapy would safely reduce the total duration of antibiotic treatment.
Inclusion Criteria:
- Adult patients (≥18 years) admitted to a critical care or intensive care unit (ICU).
- Initiation of intravenous antibiotics for suspected sepsis within 24 hours of admission, with expected continuation for at least 72 hours.
- Suspected sepsis defined as "acute organ dysfunction associated with suspected infection," assessed via the Sequential Organ Failure Assessment (SOFA) score.
Exclusion Criteria:
- Expected prolonged antibiotic therapy (>21 days).
- Severe immunocompromise unrelated to sepsis (e.g., neutropenia <500/µL).
- Anticipation of IL-6 receptor inhibitors (e.g., tocilizumab).
- Futile sepsis treatment expected to end within 24 hours.
- Declined consent or prior trial enrollment.
Intervention:
Patients were randomized into three groups (1:1:1):
- Daily PCT-guided protocol.
- Daily CRP-guided protocol.
- Standard care.
- In the biomarker-guided groups, clinicians received standardized, web-based recommendations to continue or stop antibiotics based on daily serum levels of PCT or CRP.
- The standard care group had daily blood samples but no biomarker guidance.
- The concealed design ensured clinicians were unaware of group assignments, minimizing bias.
Outcomes: Safety and Effectiveness Evaluated
Primary Outcomes:
- Total antibiotic duration (days, from randomization to day 28).
- All-cause 28-day mortality to assess safety, with a noninferiority margin of 5.4%.
Secondary Outcomes:
- Antibiotic duration and dose for the initial sepsis period.
- Unscheduled care escalation or readmission.
- Infection relapse, superinfection, and adverse reactions.
- Time to readiness for hospital discharge.
- ICU and hospital length of stay.
- 90-day all-cause mortality.

3-Results: PCT Shines, CRP Falls Short
Primary Outcomes:
- PCT-guided protocol: Significantly reduced total antibiotic duration (mean difference: 0.88 days(0.19 to 1.58) p= 0.01) and demonstrated noninferiority for 28-day mortality vs. standard care (AD: 1.57 (-2.18 to 5.32) p=0.02).
- CRP-guided protocol: No significant difference in total antibiotic duration (MD: 0.09(-0.60 to 0.79) p=0.79) and did not definitively meet the noninferiority margin for 28-day mortality (AD: 1.69 (-2.07 to 5.45) p=0.03).
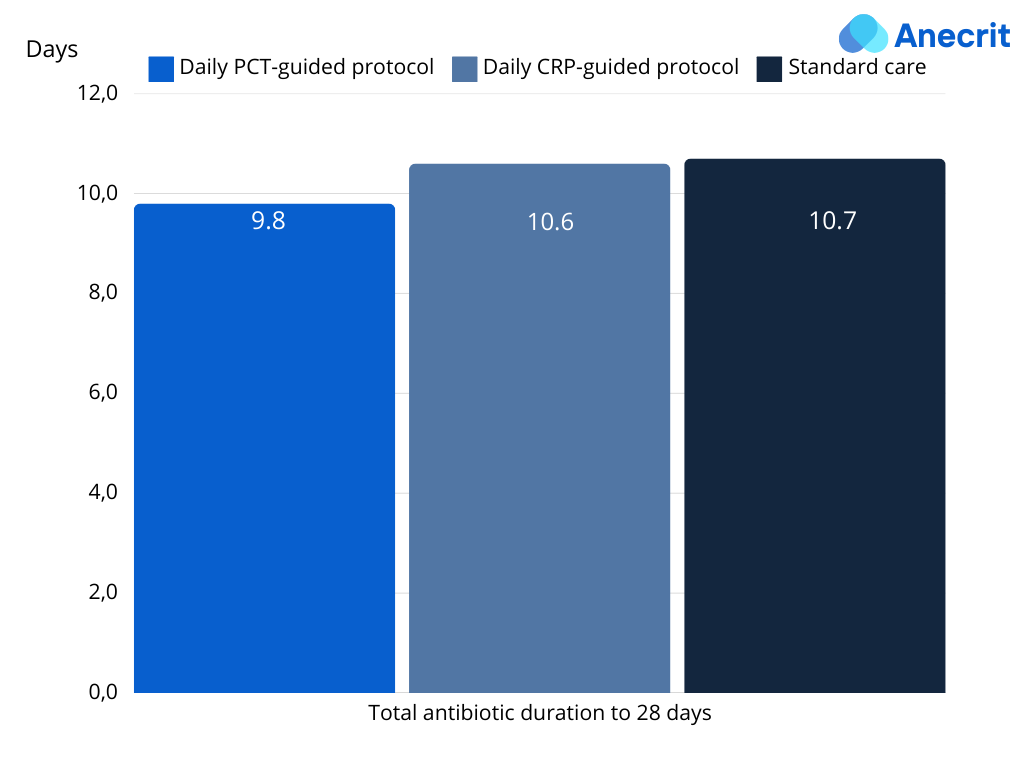
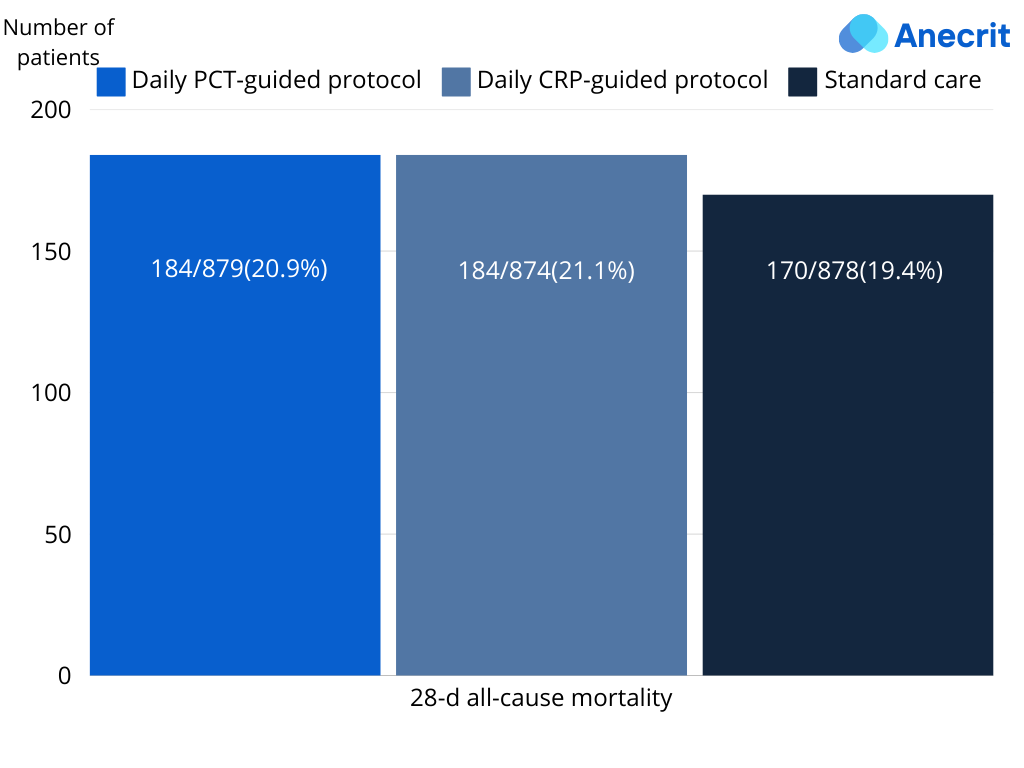
Secondary Outcomes:
- Both biomarker-guided protocols reduced antibiotic use during the initial sepsis period, though CRP's effect was not sustained through the trial.
- No significant differences in other secondary outcomes.
4-Discussion: Clinical and Practical Implications
PCT: A Promising Tool with Limitations
The ADAPT-Sepsis trial provides compelling evidence that daily PCT guidance can (safely!?) reduce antibiotic duration in critically ill adults with suspected sepsis. While a reduction of 0.88 days may seem modest, its impact on a population level could significantly curb antibiotic overuse, mitigate resistance, and minimize adverse effects. However, the cost-effectiveness of daily PCT monitoring warrants further exploration, especially in resource-limited settings where access to procalcitonin testing remains restricted.
The statement regarding the safety of the PCT-guided protocol in the ADAPT-Sepsis trial requires further nuance. While the trial met the pre-specified non-inferiority margin for 28-day all-cause mortality, this margin (5.4%) may not be sufficiently stringent to definitively rule out a small but potentially clinically significant increase in mortality associated with shorter antibiotic courses. Further investigation and longer-term follow-up are necessary to fully assess the long-term safety implications of antibiotic duration reduction guided by PCT.
CRP
The lack of effectiveness of the CRP-guided protocol underscores the importance of biomarker selection. While both PCT and CRP are inflammatory markers, PCT's more specific kinetics and reliability in bacterial infections make it superior for guiding antibiotic discontinuation.
Economic Considerations
Procalcitonin testing is costly and not universally accessible, particularly in low- and middle-income countries (LMICs). The latest guidelines from the Surviving Sepsis Campaign acknowledge these limitations. Furthermore, while some studies suggest cost savings associated with PCT-guided strategies, these benefits need validation across diverse economic contexts.
Strengths and Limitations of the Study
Strengths:
- Multicenter design enhances generalizability.
- Concealed intervention minimizes bias.
- Comprehensive outcomes robustly assess protocol effects.
- Large sample size ensures statistical power.
- Alignment with Sepsis-3 criteria strengthens clinical relevance.
Limitations:
- Potential for contamination bias due to shared care environments.
- Open CRP monitoring outside the trial protocol may have influenced decisions.
- Unadjusted multiple comparisons for primary outcomes.
- Limited applicability to resource-constrained settings.
- And as the authors mentionned the concealement strategy could have impact on the difference in the total antibiotic duration for the standard care group.
Current Guidelines and Future Directions
The 2021 Surviving Sepsis Campaign guidelines suggest using procalcitonin alongside clinical evaluation to guide antibiotic discontinuation, though this remains a weak recommendation based on low-quality evidence.
Recommendation
31. For adults with an initial diagnosis of sepsis or septic shock and
adequate source control where optimal duration of therapy is unclear,
we suggest using procalcitonin AND clinical evaluation to decide
when to discontinue antimicrobials over clinical evaluation alone.
Weak recommendation, low quality of evidence." The 2021 Surviving Sepsis Campaign guidelines p22/67 (1202).
The ADAPT-Sepsis trial bolsters support for PCT-guided strategies but highlights the need for further research to refine biomarker-driven approaches.
5-Conclusion
The ADAPT-Sepsis trial represents a potential advancement in optimizing antibiotic therapy for sepsis. However, the safety profile of the daily PCT-guided protocol warrants careful consideration. While the study met its non-inferiority margin for mortality, the pre-specified margin itself may not be sufficiently conservative to definitively exclude the possibility of a small increase in harm associated with reduced antibiotic exposure. Furthermore, challenges related to cost-effectiveness and implementation, particularly in resource-limited settings (though outside the primary scope of this trial), remain significant barriers to widespread adoption. Future research should prioritize validating these findings across diverse healthcare settings and exploring the long-term impact on patient outcomes.
6-References
- Biomarker-Guided Antibiotic Duration for Hospitalized Patients With Suspected Sepsis: The ADAPT-Sepsis Randomized Clinical Trial. JAMA. 2024 Dec 9. doi: 10.1001/jama.2024.26458.
- Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021 Nov;47(11):1181-1247. doi: 10.1007/s00134-021-06506-y.
Keywords: Sepsis, Antibiotics, Procalcitonin (PCT), C-Reactive Protein (CRP), Biomarkers, Antibiotic Stewardship, Antibiotic Duration, ADAPT-Sepsis Trial, Critical Care, Intensive Care Unit (ICU).

Comments ()